Abstract
Background:
Minimal residual disease (MRD) assessment can provide an index of robust disease control, particularly if MRD negativity is sustained over time. Whereas MRD status has well-established prognostic implications in multiple myeloma, its role in light chain (AL) amyloidosis-a related but biologically distinct plasma cell disorder-is presently under investigation.
Aims:
The aims of our study were to evaluate (1) major organ deterioration-progression-free survival (MOD-PFS) based on MRD status and (2) MRD evolution patterns during disease surveillance for patients with AL amyloidosis in hematologic complete response (hemCR) after therapy.
Methods:
We established MRD status using multiparametric 10-color flow cytometry of bone marrow aspirates. At least 2 x 10 6 events were measured using a Beckman Coulter Navios Flow Cytometer and analyzed by Kaluza Software (sensitivity level of ≤10 -5). Sequential MRD testing was performed (≥12 months apart) for patients returning for follow-up. We evaluated hematologic and organ responses or progression based on consensus guidelines (Palladini, 2012). MOD-PFS was defined as time from hemCR achievement to major organ deterioration (i.e., the kidney or heart), hematologic progression or death from any cause (whichever occurred first). Presence of MRD was not used to guide treatment decisions.
Results:
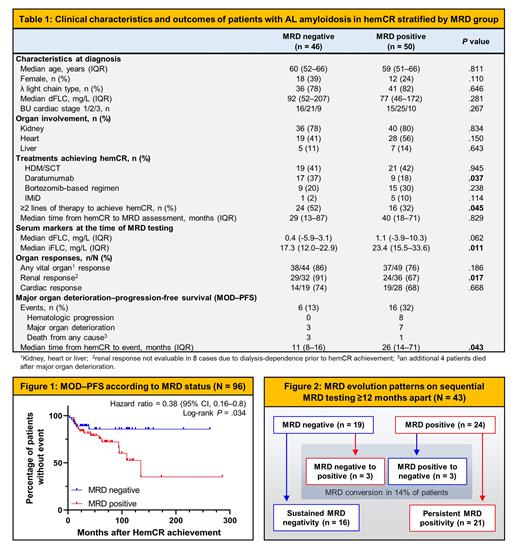
A total of 96 patients with hemCR after therapy were tested, of whom 46 (48%) were MRD negative and 50 (52%) MRD positive on first assessment. The median estimated clone size for those with MRD positivity was 3.4 x 10 -4 (range 1.4 x 10 -5 to 5.7 x 10 -3). Baseline characteristics were similar between groups, except for greater use of daratumumab and ≥2 lines of therapy to achieve hemCR in the MRD-negative group. MRD positivity correlated with a higher level of involved free light chains (iFLC) in the serum at the time of MRD assessment (23.4 mg/L vs 17.3 mg/L, P = .011). In regard to organ responses, patients with MRD negativity had a significantly higher rate of renal response (91% vs 67%, P = .017) but a similar rate of cardiac response (74% vs 68%, P = .668).
After a median follow-up of 50 months from hemCR achievement, the MRD-negative group demonstrated a superior MOD-PFS (HR 0.38, 95% CI 0.16-0.88, P = .034). Progression or organ deterioration events in the MRD-negative group occurred early after hemCR achievement, while in the MRD-positive group these events continued to occur over time. A total of 8 patients had died, including 4 in each group.
Sequential MRD testing (median 12 months apart, range 12-28 months) was performed for 43 patients, of whom 16/19 (84%) had sustained MRD negativity; 21/24 (88%) had persistent MRD positivity; and 6/43 (14%) had MRD conversion. No patient received therapy between MRD assessments. Among those with sustained MRD negativity, there were 2 major organ deterioration events. The 3 patients who experienced loss of MRD negativity (which occurred at 12, 107 and 114 months after last treatment) had no progression or organ deterioration events. Of those with persistent MRD positivity, 13 (62%) had durable hemCR and organ responses; moreover, 4/14 (29%) and 1/11 (9%) of these patients attained even deeper renal and cardiac responses, respectively, in the interval between sequential MRD testing. Clone progression (defined as >1-log growth in MRD clone size) was observed in 3/21 (14%) patients with persistent MRD positivity, none of whom demonstrated hematologic relapse.
Conclusions:
We evaluated the dynamics of MRD status in relation to clinical outcomes and found that patients with MRD negativity had longer survival free from hematologic progression, major organ deterioration or death. Longer prospective follow-up is needed to determine whether this translates into an overall survival benefit. MRD conversion occurred in a small proportion (14%) of patients, but was not linked to adverse outcomes. Understanding the role of cross-sectional MRD testing for clinical disease tracking in AL amyloidosis requires further analysis with larger sample size.
Sanchorawala: Prothena: Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Research Funding; Pfizer: Honoraria; Karyopharm: Research Funding; Sorrento: Research Funding; Celgene: Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees; Caelum: Membership on an entity's Board of Directors or advisory committees, Research Funding; Regeneron: Membership on an entity's Board of Directors or advisory committees; Proclara: Membership on an entity's Board of Directors or advisory committees; Oncopeptide: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal